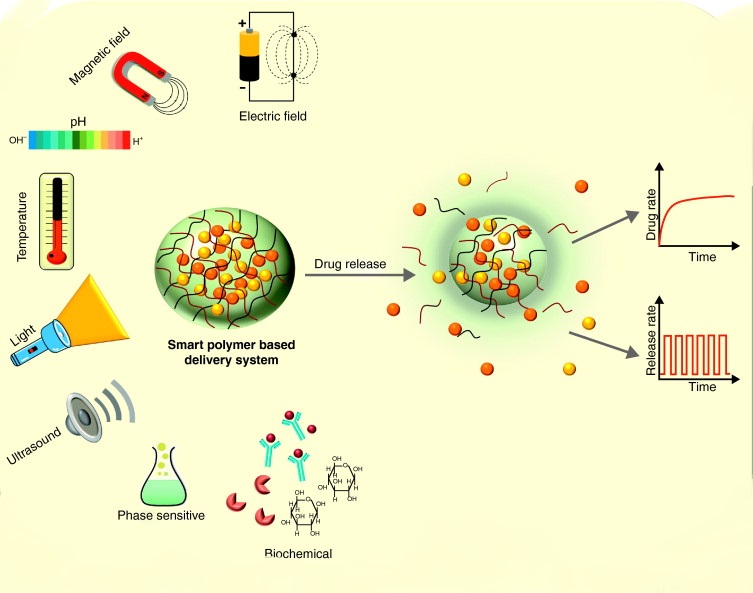
What is a smart polymer?
Inspired by the intelligent mechanisms in nature and in sync with other fields, experts have tried to make smart materials that can respond to a specific external stimulus for special and emerging applications and meet specialized needs. One of the main groups of smart materials is based on polymers because their synthesis is cheaper and easier than metals and ceramics. Smart polymers are materials that show great reversibility or physical and chemical changes under the influence of the stimulation function. These materials can respond to a stimulus or multiple stimuli such as temperature, pH, electric field or magnetic field, light intensity, organisms such as enzymes, etc., which cause macroscopic responses in the material. Most polymers can be easily functionalized using pre-polymerization or post-polymerization methods such as biological receptors. Therefore, polymer researchers have a wide range of possibilities in terms of polymer chemical structures and polymer modifications to create an unlimited number of applications for these smart materials. According to a classification, smart polymers are divided into three states based on their physical state: free chains in solution, three-dimensional gels, and surface chains. Depending on the type of response to stimuli such as change in shape or volume, the appearance of physical changes such as color change or solubility and structural changes in the polymer chain, each of these smart polymers is used in various industries such as medicine, textile, electronics, mechanics, aerospace and others. Industries have been used. These applications are progressing significantly today.
Smart block copolymers
Smart block copolymers represent a new class of functional materials with many applications in various industrial fields. Block and graft copolymers are particularly important because they generally retain the unique properties of their constituents. In comparison, random copolymers that have “average” properties of two components. Block and graft copolymers also show the way to combine two different and unusual properties in one polymer structure. The two different parts of the linking copolymers can make them with a combination of desirable properties for specific applications. Biphilic block copolymers contain at least two or more hydrophilic and hydrophobic mixed segments and supramolecular segments. Hydrophilic interactions are a fundamental driving force in the assembly of amphiphilic systems. Block copolymer micelles are generally formed by self-assembly of bivalent homopolymers or through oppositely charged surface charging in aqueous medium. Unlike small hydrophilic groups, when larger hydrophilic groups such as macromolecules are introduced, the hydrogen bonding network has no way to reorganize to accommodate large amounts of polar species. Izidak and co-workers have reported studies on temperature-responsive aqueous solutions based on poly(N,N-diethylacrylamide) and expressed changes in cloud point as a function of sodium chloride and dodecyl sulfate concentration[35]. . Tang et al have published a report on aqueous solutions of polyisopropylacrylamide to polymer concentration and molecular weight. The synthesis of hydrophobic-hydrophilic block copolymers with different clouding temperatures allows for the construction of reversible micelle-forming species.
pH responsive polymers in drug delivery systems
pH-sensitive polymers can be defined as polyelectrolytes that contain weak acidic or basic groups in their structure that accept or release protons in response to environmental pH changes. The acidic or basic groups of these polyelectrolytes can be ionized like the acidic group of monoacids or monobases; However, complete ionization of these systems is carried out due to electrostatic effects by other nearby ionized groups. This causes the apparent detection constant (Ka) to be different from that of the corresponding monoacid or monobase. Physical properties such as chain malleability, conformation, solubility, and volume of pH-sensitive polymers can cause electrostatic repulsion by making changes in the main chain or electrolyte concentration, which increases the hydrodynamic volume of the polymer.
Temperature responsive polymers
Polymers can create environmentally sensitive structures through several ways. Among the types of environment-sensitive polymers, temperature-responsive polymers have been studied more than other environment-sensitive polymers due to their many uses in the industry. Increasing attention to temperature-responsive polymers is due to their behavior in aqueous environments, which can show phase transition behavior with increasing temperature. Temperature-responsive polymers exist in various forms, including free chains in solution, cross-linked chains on a surface, networks and gels, phase-separated materials and reversible or physical networks, and discrete gels. are connected to each other.
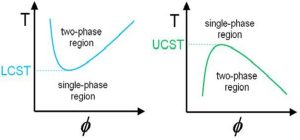
Temperature responsive polymers can show two general types of temperature behavior.
- Polymers that can adapt with increasing temperature and show the behavior of the maximum critical solution temperature (UCST).
- Polymers that become two phases with increasing temperature and exhibit the behavior of minimum critical solution temperature (LCST). Many polymers dissolve in water with increasing temperature, but these polymers become two phases with increasing temperature and create unique properties. The LCST behavior depends on the hydrogen bonding between water molecules and the structure of the monomer units in the polymer chain. Compressive changes or reversible alternating changes exist for temperature-responsive polymers at low temperature changes.
This behavior occurs due to the difference in solubility, in the phase diagrams, in the solvent/polymer binary mixture, which is accompanied by phase separation. If increasing the temperature leads to the formation of two immiscible liquid phases with different polymer concentrations, this mixture shows the lowest critical temperature of the LCST solution. It is defined as the lowest point in the binodal curve. The concentration at this minimum point is called the LCST concentration. If in the binary mixture, two liquid phases are formed by decreasing the temperature, the highest critical temperature of the solution is called UCST.



بدون دیدگاه